Butterbur Petasin Safety Profile: 24 Years of Pharmacovigilance and Causality Assessment
Between 1998 and May 2022, Butterbur Petasin exposure reached ~2.674 million patient-months worldwide, including ~2.04 million in US/Canada without any hepatobiliary AE reports in the latter region.
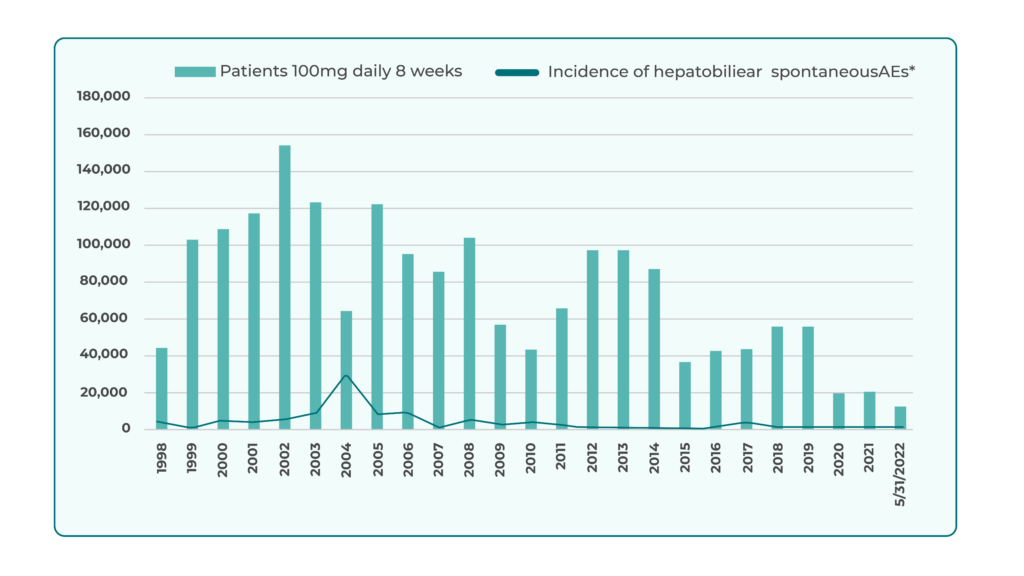
Global pharmacovigilance identified 49 spontaneous hepatobiliary cases over 24 years, corresponding to an incidence of ~1.06 cases per million 8-week treatment cycles (100 mg/day equivalent). Most showed mild, reversible liver biochemistry changes (<3 × ULN).
Over four decades, 61 total cases were documented—49 spontaneous and 12 from trials. In 10 serious spontaneous cases, causality was assessed as likely (1), unlikely (2), unclassifiable (2), and possible (5), often confounded by comorbidities or other drugs. Liver biopsies from eight cases revealed inconsistent injury patterns. Two required liver transplantation: one deemed unlikely related; the other possible but with prior drug-induced hepatitis.
Among 39 non-serious spontaneous cases, causality assessments were likely (2), unlikely (5), possible (8), unclassifiable (23), and not related (1). The 12 trial-related cases were not attributed to Butterbur Petasin. Overall, findings indicate extremely low incidence of hepatobiliary AEs, mostly mild and reversible, with no consistent hepatotoxicity pattern.
Sources
Front Neurol. 2022 Apr 26;13:864689

