Butterbur Petasin in Clinical Practice: Ideal Candidates & Contraindications
In clinical practice, Butterbur Petasin may be considered for migraine prevention in patients whose attacks significantly impair daily functioning, despite acute medication use and lifestyle adjustments.
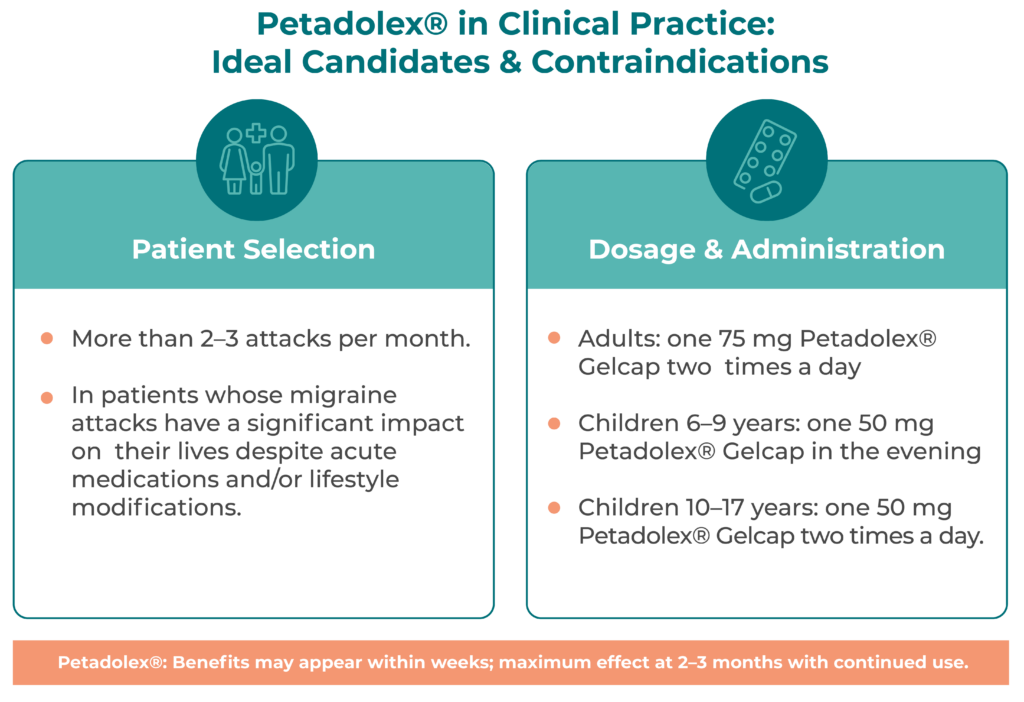
This includes individuals who continue to miss work, social engagements, or important life events. It can also be offered to patients with infrequent but severe migraines, if both patient and physician agree that preventive therapy is appropriate.
Contraindications must be carefully considered. Butterbur Petasin is not recommended in pregnant, lactating, or breastfeeding patients due to a lack of safety data. It should also be avoided in patients with liver disease. Although Butterbur Petasin undergoes purification to remove pyrrolizidine alkaloids (PAs) — compounds known to cause liver injury — precautionary avoidance in those with existing hepatic conditions minimizes potential risk.
Finally, patients with known allergies to plants in the Asteraceae, Compositae, or Daisy family should not use Butterbur Petasin. This botanical family includes ragweed, marigolds, chrysanthemums, and daisies, and allergic reactions in sensitive individuals are possible.
When used in appropriate patients, Butterbur Petasin offers a natural, evidence-based preventive option with a strong safety profile, expanding the treatment toolkit for migraine management.
Sources
Internal data available upon request

