Randomized Controlled Trials in Episodic Migraine (Pediatric)
A 2005 open-label, prospective study by Pothmann et al. evaluated Butterbur Petasin for migraine prevention in 108 children and adolescents aged 6–17 years with at least a one-year migraine history.
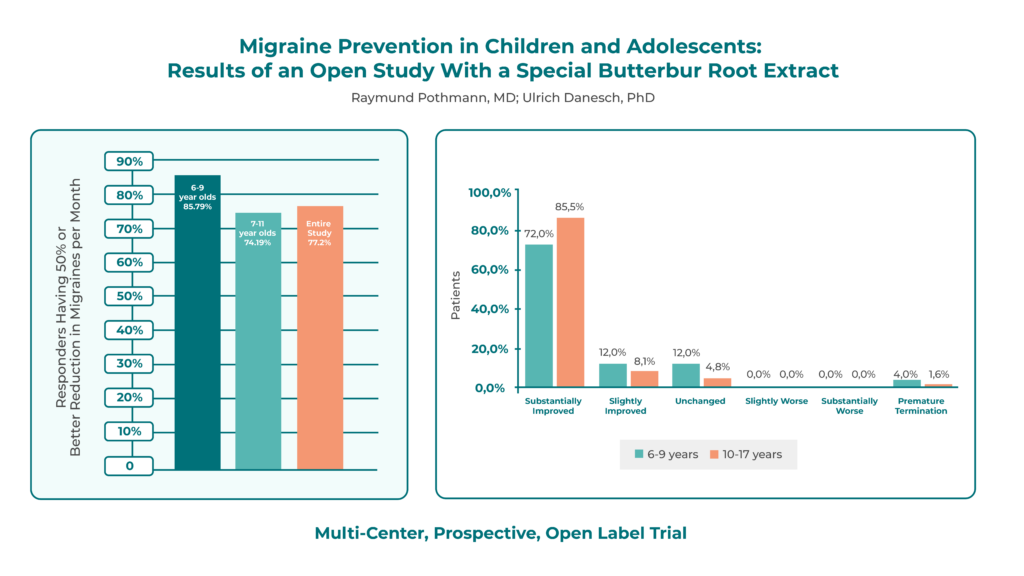
Participants received 50–150 mg daily for 4–6 months. The primary endpoint was change in migraine attack frequency.
Seventy-seven percent achieved a ≥50% reduction in attack frequency, with an overall 63% reduction from baseline. Attack duration decreased in 63% of patients, from ~10 hours to ~7 hours.
Patient-reported outcomes were strongly positive: 91% felt substantially or slightly improved, and 90% of patients and physicians noted improved well-being. Global efficacy ratings showed 72% of children aged 6–9 and 85.5% of adolescents aged 10–17 were substantially improved; 12% in both groups reported slight improvement. No patients reported worsening symptoms. Treatment discontinuation occurred in only 4% of younger and 1.6% of older participants.
Butterbur Petasin was well tolerated, with mild side effects in 7.4%—mainly burping—and no serious adverse events.
These findings, consistent with multicenter placebo-controlled trials in adults, support Butterbur Petasin as an effective and safe prophylactic option for migraine in children and adolescents.
Sources
Headache. 2005 Mar;(3):196-203

