Randomized Controlled Trial #1 in Episodic Migraine (Adult)
A 2004 randomized, double-blind, placebo-controlled trial led by Diener evaluated the efficacy of Butterbur Petasin in preventing episodic migraine.
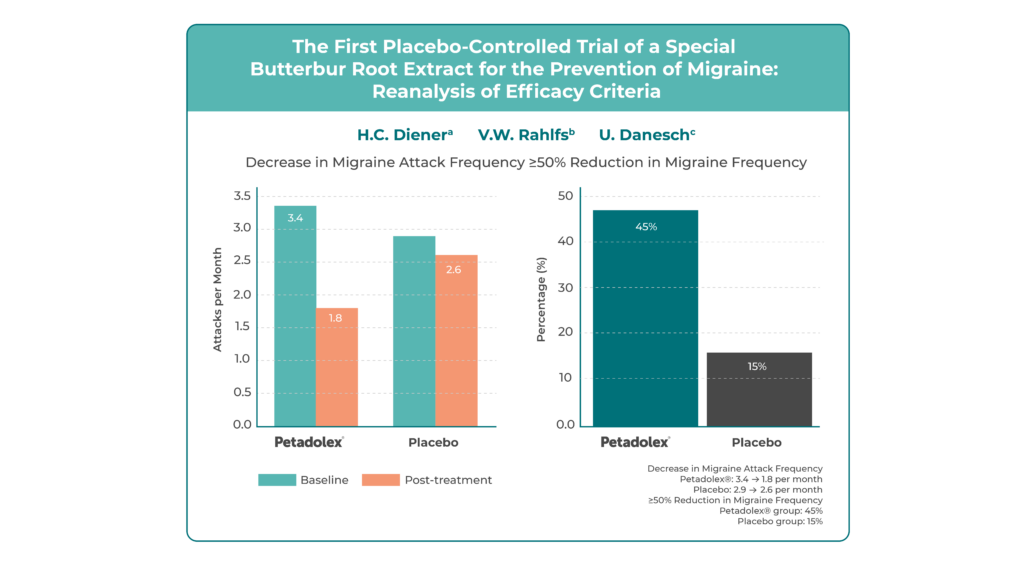
Sixty adults with at least three migraine attacks per month in the preceding three months were enrolled. Participants received Butterbur Petasin 50 mg twice daily for 12 weeks. The primary endpoint was the reduction in migraine attacks over a four-week period.
Butterbur Petasin significantly reduced migraine frequency from 3.4 to 1.8 attacks per month, compared with a smaller decrease from 2.9 to 2.6 in the placebo group (p = 0.0024). The responder rate – defined as a ≥50% reduction in attack frequency – was 45% with Butterbur Petasin versus 15% with placebo. Secondary outcomes, including number of migraine days, as well as attack duration and intensity, also showed statistically significant improvements in the treatment group.
Safety monitoring noted three patients in the Butterbur Petasin group with elevated ALT, AST, bilirubin, and erythrocyte counts. These changes were judged to have no clinical relevance. Two participants discontinued due to suspected pregnancy.
Overall, Butterbur Petasin demonstrated substantial and statistically significant benefits for migraine prevention. Nearly half of treated patients achieved a ≥50% reduction in attack frequency, and improvements were also seen across multiple headache parameters. The treatment was generally well tolerated, supporting its use as an effective and safe non-pharmacologic option for episodic migraine prevention.
Sources
Eur Neurol. 2004;51(2):89-97.

