Randomized Controlled Trial #2 in Episodic Migraine (Adult)
A 2004 randomized, double-blind, placebo-controlled trial by Lipton et al. evaluated Butterbur Petasin for episodic migraine prevention in 245 adults with at least three attacks per month.
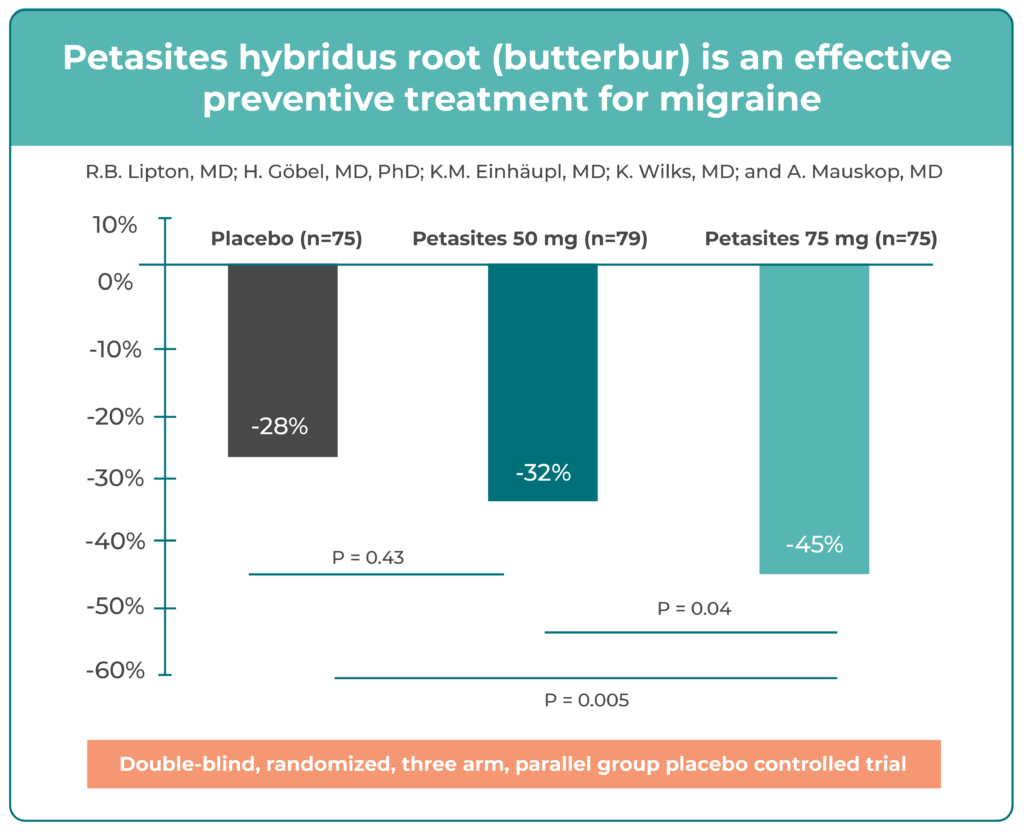
Participants were randomized to receive Butterbur Petasin 50 mg twice daily, Butterbur Petasin 75 mg twice daily, or placebo for four months. The primary endpoint was the reduction in migraine frequency.
The 75 mg twice-daily group showed the greatest benefit, with a 45% reduction in migraine frequency versus 26% for placebo. A ≥50% reduction in attack frequency (responder rate) was achieved in 68% of patients in the 75 mg group compared with 36% in the placebo group. The 50 mg group also demonstrated a reduction in migraine frequency, though the effect was smaller, indicating a dose-dependent response.
Notably, the 75 mg dose was significantly more effective than placebo as early as the first month of treatment, with benefits sustained throughout the four-month period. Secondary outcomes, including attack duration and intensity, also favored the higher dose.
Adverse events were generally mild, with gastrointestinal discomfort—particularly burping—being the most common. No serious treatment-related adverse events were reported.
This trial confirmed the efficacy of Butterbur Petasin for migraine prevention, with clear evidence of a dose-response relationship and an early onset of benefit. The 75 mg twice-daily regimen provided the most robust and sustained improvement, was well tolerated, and offers a clinically meaningful preventive option for adults with episodic migraine.
Sources
Neurology. 2004 Dec 28;63(12):2240-4

