Migraine Epidemiology, Diagnosis, and Preventive Gaps
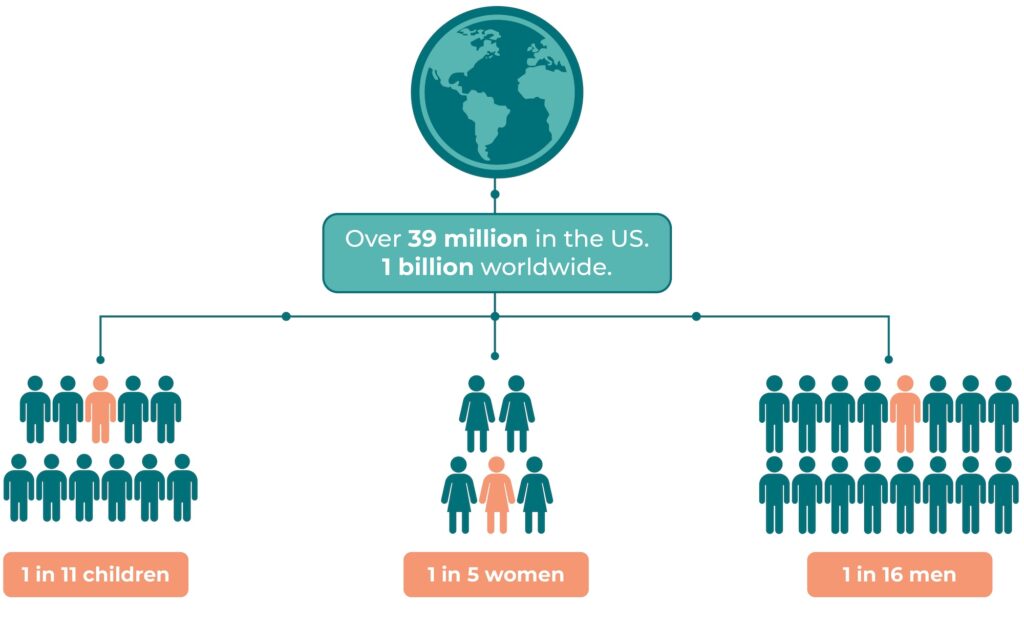
Migraine is a highly prevalent and disabling neurological disorder affecting over 1 billion people globally, and more than 39 million individuals in the United States.
It is the second leading cause of years lived with disability and disproportionately affects women and adolescents.
Despite its impact, migraine remains underdiagnosed and undertreated, particularly in primary care settings.
The International Classification of Headache Disorders (ICHD-3) provides formal criteria for diagnosis, requiring ≥5 attacks of unilateral, pulsating headache lasting 4–72 hours with associated nausea and/or sensory sensitivity.
Clinical tools like the ID-Migraine™ screener enable fast and accurate recognition: patients answering “yes” to 2 of 3 core symptoms (nausea, photophobia, functional disability) have a 93% likelihood of migraine. Preventive treatment is indicated for patients with ≥4 monthly headache days or significant disability. However, many standard preventive agents (e.g., beta blockers, antidepressants, anticonvulsants) suffer from poor adherence due to tolerability and safety concerns.
Calcitonin Gene-Related Peptide (CGRP)-targeting treatments—such as erenumab, galcanezumab, fremanezumab, eptinezumab, atogepant, and rimegepant—have emerged as effective and well-tolerated options for migraine prevention.
However, their widespread use remains limited by high cost, restricted insurance coverage, and a lack of long-term safety data beyond seven years. In contrast, nutraceuticals like magnesium, CoQ10, riboflavin, and butterbur offer a more accessible and affordable approach, with a favorable safety profile and strong consumer preference for non-pharmacologic therapies.

Sources
- Lancet Neurol. 2018;17:954-976
- Neurology. 2007;68:343-9
- Cephalalgia. 2018;38:1-211; J
- Headache Pain. 2018;19:77
- Cephalgia. 2016:1112-1133

