Safety Profile – Preclinical Toxicity Studies
Chronic Toxicity in Rats (6 months): No evidence of liver damage was observed. Apart from a ~2-fold increase in bilirubin, liver function tests (LFTs) showed insignificant or minimal changes. All LFTs normalized during ongoing treatment and after a 4-week recovery period. Minor biliary duct changes at high doses were interpreted as metabolic adaptation rather than toxicity.
Mini-Pig Study (28 days): (Not shown in slide): No liver toxicity or enzyme changes were detected. Liver histopathology revealed no structural damage.
In Vitro Studies: (Not shown in slide): Human and rat liver cell models showed no significant cytotoxicity or changes in liver enzyme activity.
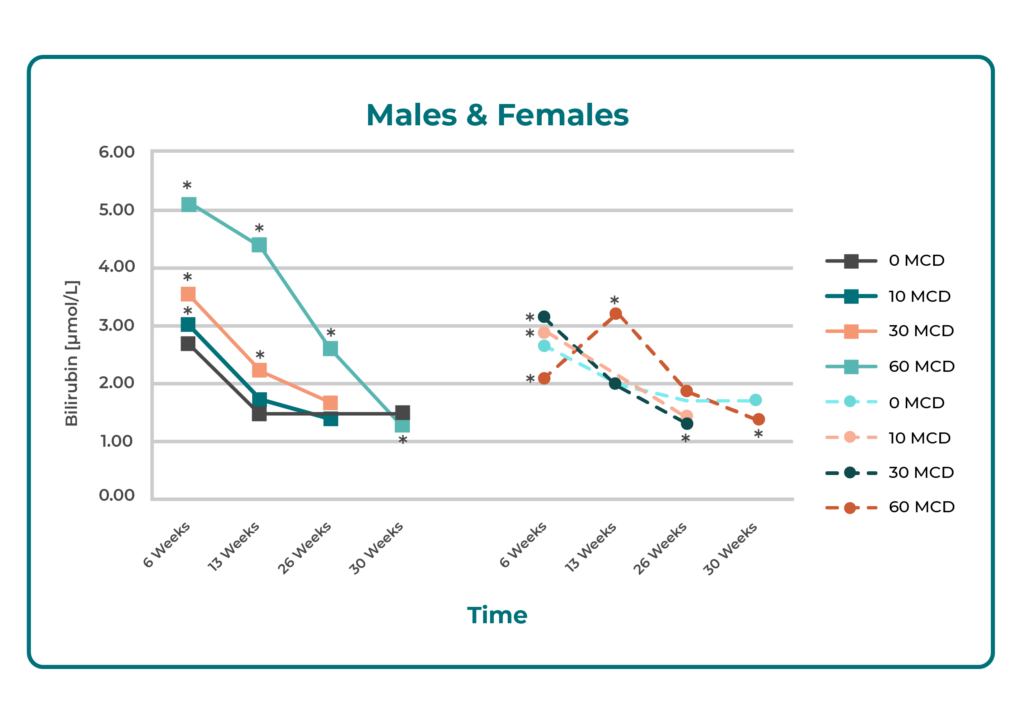
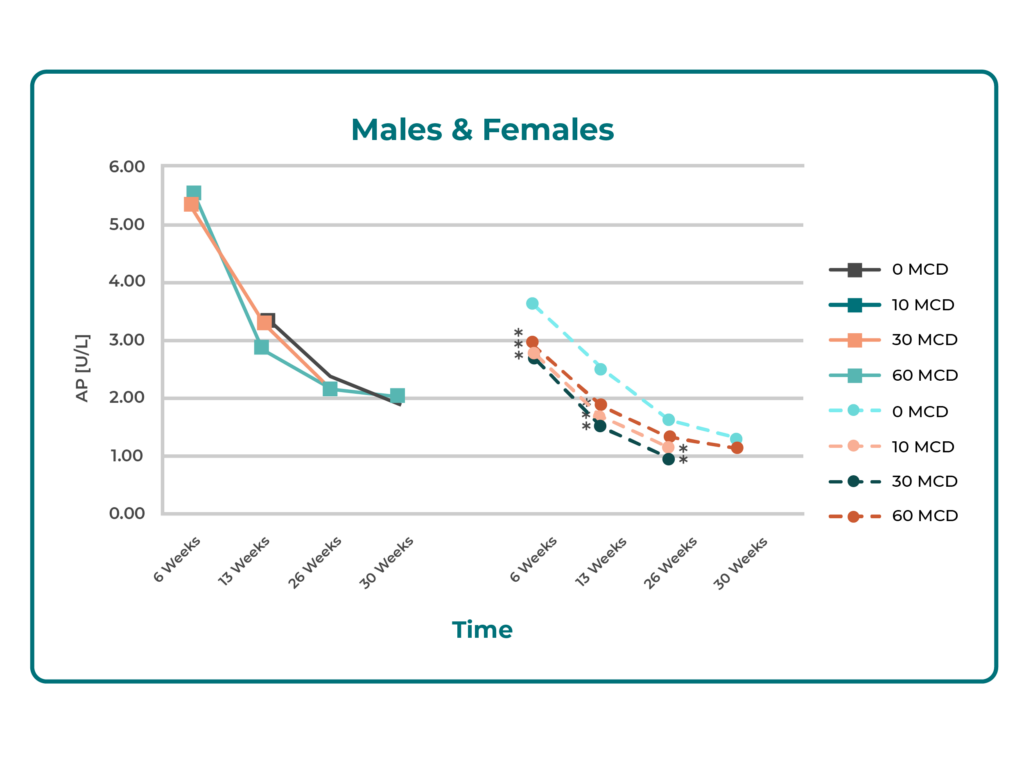
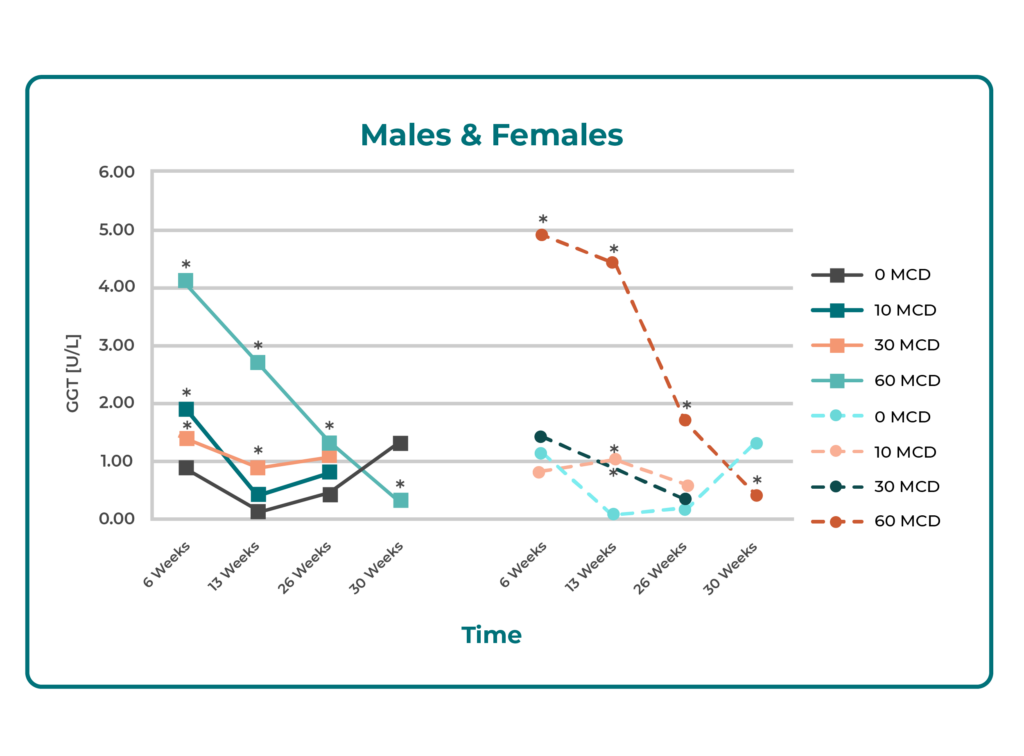
Summary: Across animal and in vitro models, Butterbur Petasin demonstrated no clinically relevant hepatotoxicity, even with prolonged or high-dose exposure. Observed changes were minimal, reversible, and consistent with physiological adaptation rather than toxic injury.
Sources
Front Neurol. 2022 Apr 26;13:864689

